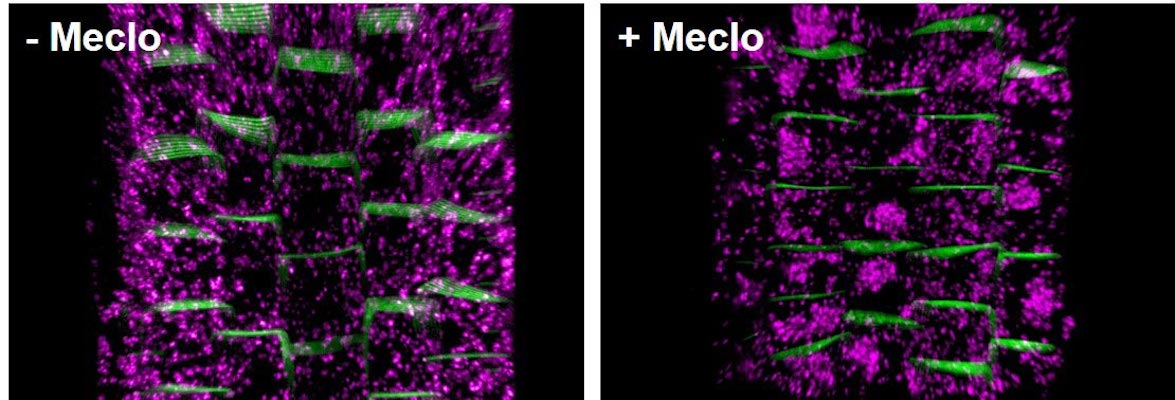
The 3D structures of these large protein assemblies – the first described for any plant species – are a step towards being able to develop improved herbicides that target plant respiration. They could also aid the development of more effective…
Read More