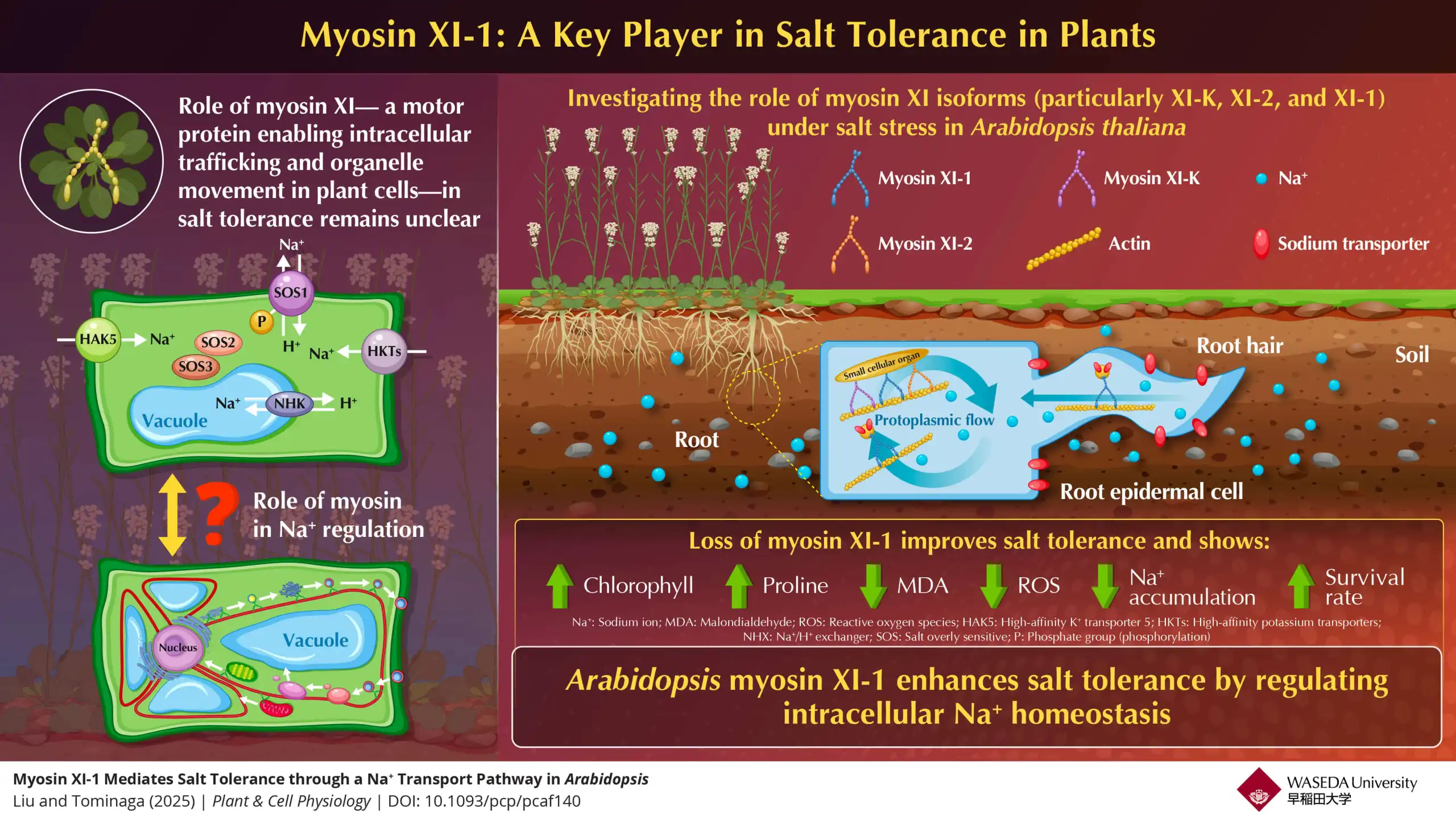
Salt stress is a major factor that impairs plant growth and yield. Researchers have revealed that Arabidopsis thaliana myosin XI-1 significantly regulates adaptation to salt stress via intracellular sodium ion (Na⁺) homeostasis. It can thus serve as a promising molecular…
Read More