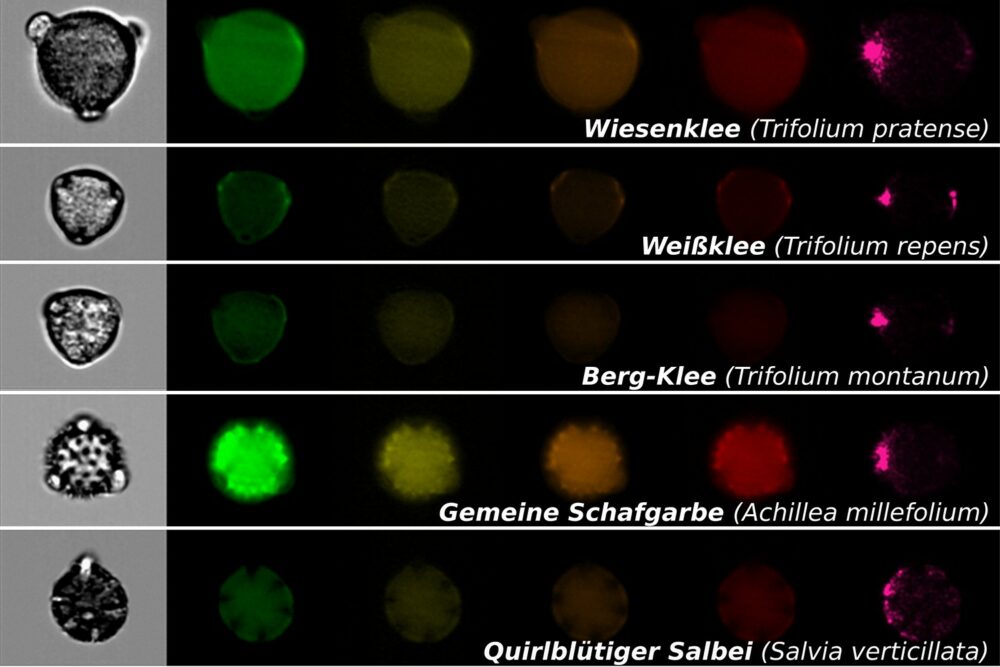
Researchers found that honey bee apiaries and urban areas reduce populations of certain native bees, particularly ground-nesters and late-season foragers. Six of 33 studied bee genera were affected, notably Svastra and Florilegus. Strategies like planting late-blooming flowers and preserving ground…
Read More