This week we spoke to Dr. Winfried S. Peters from Indiana University/Purdue University Fort Wayne (IPFW). His research mainly focuses on the biomechanics of plant cells, which led him to take a second look at some of the ideas of botanists in the 19th and early 20th century and use modern techniques to make exciting new discoveries.

Dr Winfried S. Peters, Indiana University/Purdue University Fort Wayne (IPFW), next to several tons of land-plant sieve elements!
Could you begin by describing your research interests?
I am interested in the biophysical aspects of the physiology of plants and animals. In plants, my research focuses on the mechanics of growth and morphogenesis, and on the cell biology of long-distance transport in the phloem. For both topics, a solid background in the history of the field can be quite helpful – I love studying the old literature to reconstruct the ideas botanists had a century or two ago regarding the functioning of plants.
At the recent New Phytologist Symposium, entitled “Colonization of the terrestrial environment 2016”, you presented fascinating work on the sieve tubes of kelp, which resemble the phloem tubes of vascular plants. What is the purpose of these tubes?
In large photosynthetic organisms, not all parts of the body are truly autototrophic. Some tissues produce more material by photosynthesis than they need, while others produce less than they require or none at all– think of green leaves and growing root tips. Over-producing tissues can act as sources and export photoassimilates to needy sink tissues. Sieve tubes are arrays of tubular cells that mediate this exchange, enabling the rapid movement of photosynthate-rich cytoplasm between sources and sinks.
What techniques did you utilize to investigate the function of these tubes, and what did this reveal?
During my recent sabbatical, I became involved in this project in the lab of my friend and long-term collaborator, Professor Michael Knoblauch. Michael heads the Franceschi Microscopy and Imaging Center at Washington State University, where we studied sieve tubes of the Bull Kelp (Nereocystis luetkeana) using a variety of state-of-the-art microscopy techniques. Most importantly, we employed fluorescent dyes to visualize transport in sieve tube networks. To do this, one needs to work with intact kelp, which is demanding given a thallus size of 12 meters and more. So we moved to Bamfield Marine Sciences Centre on Vancouver Island, where Bull Kelp is a ‘common weed’.
A particularly important result was the pressure-induced reversal of the flow direction in sieve tubes and across sieve plates. This was in line with Ernst Münch’s (1876-1946) theory, who suggested that sieve tube transport was driven by osmotically generated pressure gradients.

An intact Nereocystis luetkeana is kept in a tank (right) while sieve tube transport is studied using a fluorescence microscope. Photo credit: Michael Knoblauch.
How do the biomechanics of the kelp sieve tubes differ from the phloem tubes of higher plants?
Regarding cytoplasmic translocation, there doesn’t seem to be a difference – in higher plants as in kelps, the contents of the sieve tubes move in bulk flow – but wounding responses differ drastically. After wounding, we found that kelps have a massive swelling of the walls, which reduced the sieve tube diameter by more than 70%. By injecting silicon oil into severed kelp sieve tubes we demonstrated that wall swelling was fully reversible, and that the swelling state of the walls depended on intracellular pressure.
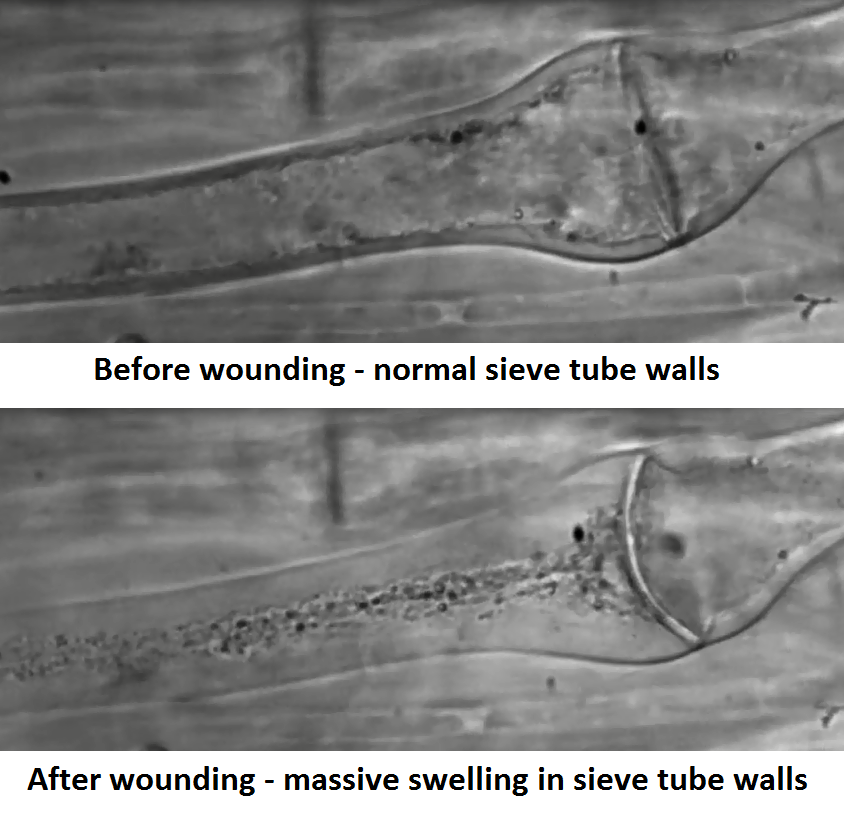
Sieve wall tubes swell after wounding due to changes in intracellular pressure. (Images taken from video below).
Have reversible wall-swelling reactions been observed in other species, and what are the implications of this finding?
We have observed the wall-swelling response in all kelp species examined. Ironically, there is no shortage of drawings and photographs of kelp sieve tubes with swollen walls in the literature over the last 130 years; however, the dynamics of cell behavior remained hidden in plain sight because fixed tissue samples rather than fully functional, whole organisms were studied. Consequently, sieve tubes with swollen walls were misinterpreted as senescent cells. There also are publications on turgor-dependent cell wall swelling in red and green algae, but these ceased around 1930.
Afterwards, wall swelling was completely forgotten, judging from the textbooks. This is remarkable, as Wilhelm Hofmeister (1824-1877), often celebrated as a founding father of plant biomechanics, denied a significant role for osmotic processes in the generation of turgor, the hydrostatic pressure within plant cells. Rather, he maintained that living cells were pressurized by the swelling of their walls. The example of the kelp sieve tube shows how easy it is to remain unaware of wall swelling when it happens right before our eyes. Maybe we should take Hofmeister’s idea seriously once again?
What are the evolutionary implications of your work?
Brown algae and vascular (land) plants are only remotely related, and their sieve tube networks certainly evolved independently of each other. It seems surprising that such sophisticated structures, which serve a complex function that integrates the physiology of the entire organism, have evolved at least twice, but think again. Real cells are not embedded in a totally homogeneous environment, and neither is the cytoplasm within the cell a homogeneous solution. Thus every cell experiences gradients of solute concentrations along its inner and/or outer surface. As a consequence, differential water fluxes across the plasma membrane will occur, resulting in movements of the cell contents. In other words, Münch flow, the cytoplasmic bulk flow driven by osmotically generated pressure gradients, is not a peculiar process operating specifically in sieve tubes, but a ubiquitous phenomenon. Sieve tubes consist of cells that simply do the things cells do, just a little more efficiently as usual. In this view, the repeated convergent evolution of sieve tube networks is not really unexpected.
But kelps resemble land plants in other ways too. As in land plants, kelp cell walls are made of cellulose (at least partly), kelp cells are connected through plasmodesmata, and the kelp life-cycle is a sporophyte-dominated alternation of generations. Evidently, none of these features represents a specific adaptation to life on dry land.
If you’d like to know more about this fascinating work, it was been published in the following articles:
Knoblauch, J., Peters, W.S. and Knoblauch, M., 2016. The gelatinous extracellular matrix facilitates transport studies in kelp: visualization of pressure-induced flow reversal across sieve plates. Annals of Botany, 117(4), pp.599-606.
Knoblauch, J., Drobnitch, S.T., Peters, W.S. and Knoblauch, M., 2016. In situ microscopy reveals reversible cell wall swelling in kelp sieve tubes: one mechanism for turgor generation and flow control? Plant, Cell and Environment, 39(8), pp.1727-1736.






