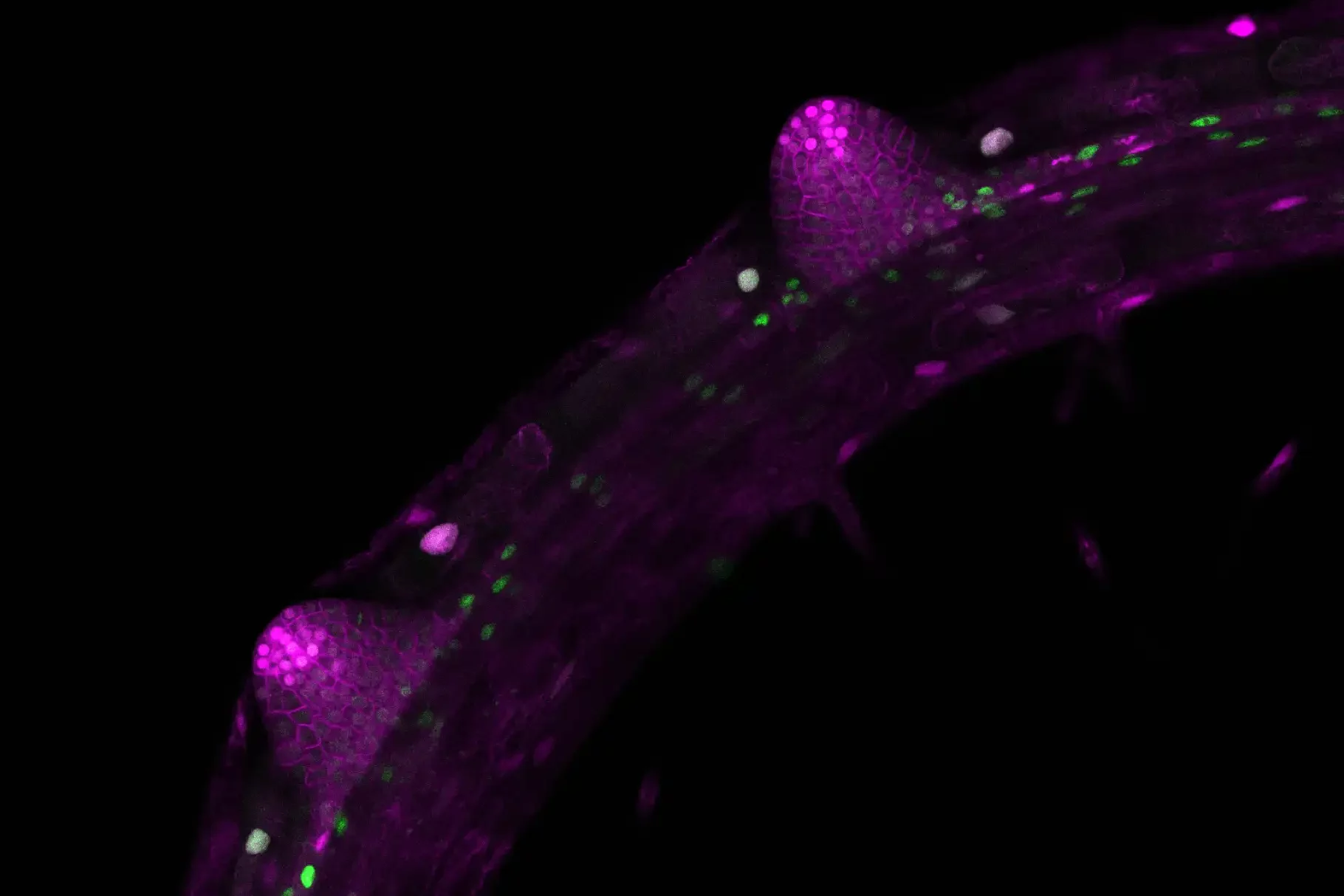
Biologists identify the molecular mechanism that controls root branching
Along with sugar reallocation, a basic molecular mechanism within plants controls the formation of new lateral roots. An international team of plant biologists has demonstrated that it is based on the activity of a certain factor, the target of rapamycin (TOR) protein. A better understanding of the processes that regulate root branching at the molecular level could contribute to improving plant growth and therefore crop yields, according to research team leader Prof. Dr Alexis Maizel of the Centre for Organismal Studies at Heidelberg University.
Good root growth ensures that plants can absorb sufficient nutrients and grow, thus contributing to their general fitness. To do that, they must align the available resources from metabolic processes with their genetic development programmes. Plants bind carbon dioxide (CO2) from the atmosphere in their leaves and convert it to simple sugars via photosynthesis. In the form of fructose and glucose, these simple sugars are also allocated in the roots, where they drive the growth and development of the plant.
Prof. Maizel’s team used the thale cress Arabidopsis thaliana, a model plant in plant research, to study how this process occurs at the molecular level. Their investigations focus on what role glucose plays in forming lateral roots. “We do know that, besides plant hormones, sugar from the shoot is also allocated in the roots, but how the plant recognises that sugar resources are available for forming lateral roots has not been understood thus far,” explains Dr Michael Stitz, a researcher on Alexis Maizel’s team.
The studies at the metabolism level showed that Arabidopsis forms lateral roots only when glucose breaks down and carbohydrates are consumed in the pericycle – the outermost cell layer of the main root cylinder. This process is controlled at the molecular level by the target of rapamycin protein. This factor controls critical signal networks and metabolic processes in plants as well as in animals and humans. Its activity is governed by the interaction of growth factors like the plant hormone auxin and nutrients like sugar.
Using Arabidopsis, the researchers discovered that TOR becomes active in the pericycle cells only when sugar is present there. So-called founder cells then form the lateral roots through cell division. Prof. Maizel: “TOR assumes a kind of gatekeeper role; when the plant activates the genetic growth programme responsible for root formation via the hormone auxin, TOR checks whether there are sufficient sugar resources available for this process.” TOR acts by controlling the translation of specific auxin-dependent genes, blocking their expression if there aren’t sufficient sugar resources available. When the researchers suppressed TOR activity, no lateral roots were formed. “That suggests that a fundamental molecular mechanism is involved,” states the Heidelberg plant biologist.
At the same time, the researchers demonstrated that TOR controls, via a similar mechanism, the formation of roots from other plant tissues – the so-called adventitious roots. According to Prof. Maizel, the results from their investigations could also be of interest for agricultural applications. “They could potentially be used to develop new strategies for plant growth optimised for various environmental conditions and better crop yields,” emphasises the researcher.
The research was funded by the German Research Foundation. In addition to the plant biologists from the Centre for Organismal Studies of Heidelberg University, researchers from the University of Strasbourg (France), the Max Planck Institute of Molecular Plant Physiology in Potsdam, the Centre for Research in Agricultural Genomics in Barcelona (Spain), and University College Cork (Ireland) also contributed. The results of their research were published in “The EMBO Journal”.
Read the paper: The EMBO Journal
Article source: Heidelberg University
Image: Two lateral root primordia develop from the main root of Arabidopsis thaliana. The images (with false colours) were taken with a confocal microscope. Credit: Michael Stitz, Heidelberg University