



Akiko Nakazaki, PhD student at Kyoto University
Kon-nichiwa! (Hello!) I am Akiko Nakazaki, a PhD student studying plant molecular cell biology at Kyoto University in Japan.
I’m interested in plant defense – specifically the glucosinolate-myrosinase defense system, which is specific to Brassicales such as Arabidopsis thaliana. Glucosinolates are a group of secondary metabolites stored in separate cells to myrosinases, the enzymes that break them down. Upon tissue damage, the glucosinolates and myrosinases are released from their cells and combine. The glucosinolates are hydrolyzed to volatile repellent compounds such as isothiocyanates and nitriles.
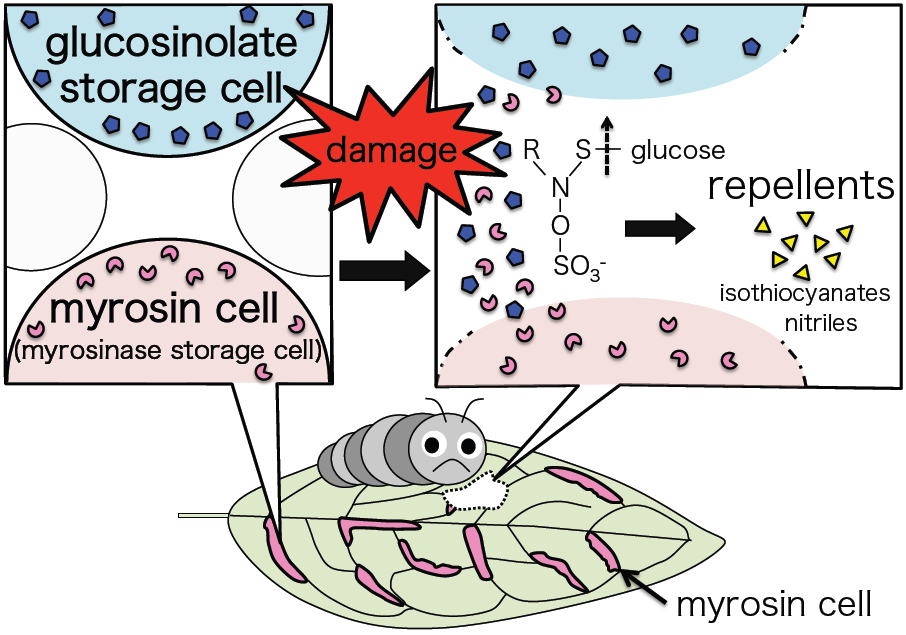
When damaged, cells containing glucosinolate and myrosinase are ruptured, releasing their contents. The glucosinolate is broken down by the myrosinase into volatile compounds that repel herbivores
I was impressed by this ingenious and rational survival strategy! I want to reveal this defense system at the cellular level, and am researching it in Arabidopsis thaliana by performing microscopic observations, bioassays with insects, and so on.
Are you interested in how PhD students from other countries spend their day in the laboratory? I am! Let me tell you about my typical day in the lab.
I wake up at 8:30am, and have morning coffee and toast for breakfast while reading a newspaper. Then, I get dressed and ride on my bicycle to the University. During the ride (about 10 minutes), I remind myself of the day’s schedule. I get to the lab at 10am and take my seat. All the members of the lab have their own desk and workbench. I turn on my computer and check my emails.
In the daylight, I basically do experiments and read papers. I start doing microscopic observations and lose track of time until I hear my stomach growling and realize that it is almost 2pm. I have lunch at the eating space in lab. In this room, there are always some lab members who are eating, discussing their research, playing social games, etc. After lunch, I report the result of my microscopic observations to my boss and we have a brief discussion about it.
Then, I return to my seat and realize the primers I ordered yesterday have arrived. I perform a PCR and prepare an agarose gel for electrophoresis. While I am waiting for the PCR to end, I search PubMed and Google Scholar for new papers to read. I load the PCR products to the gel and check that the PCR worked. In the evening, I allocate myself free time for doing more experiments, reading more papers, preparing research presentations, discussions, etc.
I’ve sought a more effective way to advance my research through trial and error. For example, when I started researching in the lab I was a little too ambitious, and planned my schedule too tightly. I sometimes felt tired and depressed when my research was not right on schedule, as is often the case. In these negative moods I couldn’t enjoy my work, so I adopted a schedule with more free time. Because of this change, I’ve come to be able to work flexibly and keep a positive frame of mind.
I’m home between 10pm and midnight. At home, I have a late dinner and take a good long soak in the bath (my favorite time of day!). I go to bed at 2am.
On weekends I enjoy playing badminton, learning traditional Japanese dance and shopping. I try to make plans without lab work as much as I can, however I’m not able to do avoid it sometimes when I am struggling to get new data before academic conferences and progress reports. Leaving the lab allows me to get rid of stress and feel refreshed for a healthy next week. Furthermore, I devise ways to work more efficiently on weekdays, because I am required to take time off at the weekends.
My boss always says, “It is important to value encounters with people and things.” It wasn’t until recently that I finally understood that message! I have found that experiments may not always work well, but when I look at it from a different angle, even experiments that haven’t gone the way I’d wanted could make me aware of something new and interesting. This awareness could also be brought about through discussions with others.
I am grateful for being able to receive this opportunity. Thank you.
Akiko Nakazaki is in the first year of her doctoral program in the Department of Botany, Graduate School of Science, Kyoto University, Japan.

Zostera marina. Public domain, via Wikimedia Commons.
It’s the ancient story of plant evolution: photosynthetic algae moved to damp places on land, eventually evolving more complex architecture, and spreading across almost all terrestrial habitats. To cope with the drier conditions, plants developed roots to absorb water, and vascular tissue to transport it; a waxy cuticle coating their surfaces to prevent evaporation; and microscopic pores called stomata that open to allow carbon dioxide to diffuse in for photosynthesis but close to prevent excessive water loss.
How, then, does eelgrass (Zostera marina) fit in to this tale? It’s a monocot descended from the flowering plants, but it has turned its back on dry land and returned to the sea; a rare feat that only appears to have happened on three occasions. The recent sequencing of the eelgrass genome has revealed several interesting insights into the dramatic genetic changes that have allowed it to adapt to what lead author Professor Jeanine Olsen described as, “arguably the most extreme adaptation a terrestrial (and even a freshwater) species can undergo.”
Sayonara to stomata
If you live in the sea, conserving water isn’t your main concern. Eelgrass was known to lack stomata, but genetic comparisons to other species, including its freshwater relative Spirodela polyrhiza, revealed the first surprise of the study: eelgrass has lost not only its stomata but also the genes involved in their development and patterning. “The genes have just gone, so there’s no way back to land for seagrass,” said Olsen.
A difference in defense
When angiosperms are attacked by herbivores or pathogens, their defense response typically involves the release of volatile secondary metabolites through their stomata. How can eelgrass release these compounds without stomata? The answer is: it doesn’t. The genome study found that eelgrass is missing crucial genes involved in making ethylene (an important hormone release in times of stress), as well as those responsible for producing non-metabolic terpenoids, which act to repel pests.
Selective pressures of the marine environment differ greatly from those of terrestrial habitats, so different pathways may be involved. Second, eelgrass has a wide repertoire of pathogen resistance genes, which suggests that it is exposed to a very different set of pathogens that may not respond to typical immune responses. Third, volatile secondary metabolites are often involved in attracting pollinators; this is not believed to be necessary in eelgrass, where submarine pollination occurs using the water itself.

Zostera marina – National Museum of Nature and Science, Tokyo. Public domain, CC0 1.0, via WikiMedia Commons.
Changing the cell wall
Eelgrass is subject to extremely salty conditions, and it’s had to adapt to osmotic stress. Unlike typical plant cell walls, eelgrass has engineered its cell wall matrix to retain water in the cell wall, even during low tide. This involves depositing sulfated polysaccharides and low methylated pectins in the cell wall matrix, but until its genome was sequenced no-one knew exactly how. It turns out that eelgrass has rearranged its metabolic pathways: “They have re-engineered themselves,” Olsen explains.
Living with a lack of light
Some species of Zostera can grow in water 50m deep, where light levels are reduced and shifted into a narrow wavelength range; ultraviolet (UV), red and far-red light have particularly low penetration after the first 1–2m of seawater. In a classic eelgrass ‘use it or lose it’ response, it has lost the UVR8 gene, which is responsible for sensing and responding to UV damage, as well as the phytochromes associated with red and far-red receptors. It does, however, retain the photosynthetic machinery, including photosystems I and II.
Unravelling angiosperm evolution
The recent eelgrass publication has revealed how this plant has either lost or adapted typical angiosperm traits to suit its needs, by ditching its stomata, volatile secondary metabolites and certain light sensing genes, or by altering the structure and function of the cell wall. It also developed adaptations that enable gas exchange, help pollen stick to submerged stigmas, and promote nutrient uptake.
Could these adaptations be useful in crop breeding? While a lack of defense compounds would probably be a step backwards, it would be extremely useful to understand how eelgrass copes with biotic stresses without them. Removing light receptors would also be problematic, but could eelgrass help us to develop crops that can grow in shaded conditions, perhaps in intercropping systems? What can we learn from eelgrass’ nutrient uptake and salt-tolerant adaptations?
Now that we have seen some of the secrets of eelgrass, how can we best make use of them?
Read the paper: The genome of the seagrass Zostera marina reveals angiosperm adaptation to the sea (Open Access)
Read the editorial: Genomics: From sea to sea (paywall)
Read the press release: Genome of the flowering plant that returned to the sea